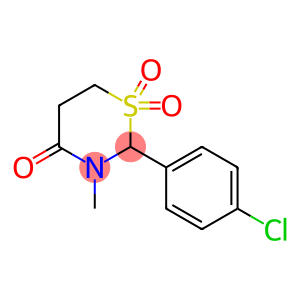
2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-dioxide
chlormezanone
CAS: 80-77-3
Molecular Formula: C11H12ClNO3S
2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-dioxide - Names and Identifiers
2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-dioxide - Physico-chemical Properties
| Molecular Formula | C11H12ClNO3S |
| Molar Mass | 273.74 |
| Density | 1.2205 (rough estimate) |
| Melting Point | 114 °C |
| Boling Point | 534.5±50.0 °C(Predicted) |
| Flash Point | 277.1°C |
| Solubility | DMSO 55 mg/mL;Water <1 mg/mL;Ethanol 26 mg/mL. Slightly soluble in water or ethanol, easily soluble in acetone, chloroform, insoluble in benzene. |
| Vapor Presure | 1.68E-11mmHg at 25°C |
| Appearance | White crystalline powder |
| Color | White to Pale Yellow |
| Merck | 14,2106 |
| pKa | -2.37±0.40(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.6300 (estimate) |
| MDL | MFCD00143951 |
| Physical and Chemical Properties | White crystalline powder. Melting point 116.2-118.2 °c. Slightly soluble in water or ethanol, soluble in acetone, chloroform, insoluble in benzene. Slightly odorous. Decomposition upon heating in an inorganic acid or base. |
| Use | Weak tranquilizer, used for nervous tension fear, chronic fatigue, Anxiety, excitement and irritability caused by certain diseases such as insomnia |
| In vivo study | Comparative results obtained with Chlormezanone in the dosage levels capable of producing more than 80% depression in the contralateral extensor. |
2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-dioxide - Risk and Safety
| RTECS | XJ1050000 |
2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-dioxide - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| biological activity | Chlormezanone is a non-benzodiazepine for the treatment of anxiety disorders. It has been suggested for the treatment of muscle spasms. |
| Target | Benzodiazepine |
| Use | is used as a weak neuroleptic. With sedation, stability and central muscle relaxation effect. Can also be used for nervous fear, chronic fatigue, Anxiety, excitement, and certain diseases caused by irritability and insomnia. Because of muscle relaxation, with analgesics can be used for back acid, hard neck, bone pain, spine and limbs pain and rheumatism Arthralgia. A weak neuroleptic, used for nervousness, fear, chronic fatigue, Anxiety, agitation and irritability caused by certain diseases such as insomnia |
| production method | from p-chlorobenzaldehyde by condensation with methylamine solution, mercaptopropionic acid cyclization, potassium permanganate solution oxidation. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 21:54:55
Supplier List
Multiple SpecificationsSpot supply
Product Name: Chlormezanone Visit Supplier Webpage Request for quotationCAS: 80-77-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Chlormezanone Visit Supplier Webpage Request for quotationCAS: 80-77-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Chlormezanone Request for quotation
CAS: 80-77-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 80-77-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple SpecificationsSpot supply
Product Name: 2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-Dioxide Visit Supplier Webpage Request for quotationCAS: 80-77-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Chlormezanone Visit Supplier Webpage Request for quotationCAS: 80-77-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Chlormezanone Visit Supplier Webpage Request for quotationCAS: 80-77-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Chlormezanone Request for quotation
CAS: 80-77-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 80-77-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Multiple SpecificationsSpot supply
Product Name: 2-(4-Chlorophenyl)tetrahydro-3-methyl-4H-1,3-thiazin-4-one 1,1-Dioxide Visit Supplier Webpage Request for quotationCAS: 80-77-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



